Calculate Kp for the below reaction at 25°C:
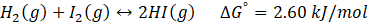
Solution:
Interpretation: We will use the equation that describes the relation between equilibrium constant (Kp ) and Gibb’s free energy change (ΔG°).
ΔG° = – RTlnKp
Where, ΔG° = Standard free energy change
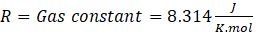
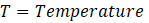
Kp = Equilibrium constant in terms of pressure
Given: ΔG° = 2.60 kJ/mol
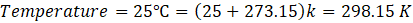
Solution: ΔG° = – RTlnKp
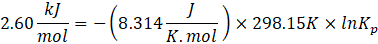
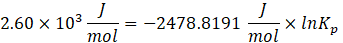
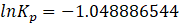
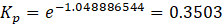
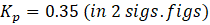
Therefore, Kp for the reaction, H2 (g) + I2 (g) ⇄ 2HI (g) is 0.35 .
