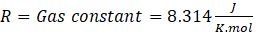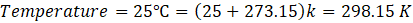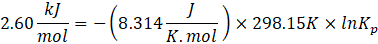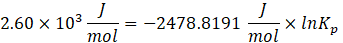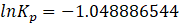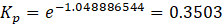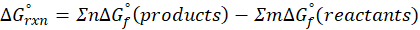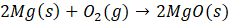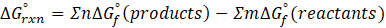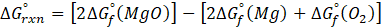For the autoionization of water at 25°C,
H2O (l) ⟺ H+(aq) + OH– (aq)
kw= 1.0 × 10-14 . What is ΔG° for this process?
Interpretation
The autoionization of water is the process in which a small fraction of water molecules transfer a proton to one another, producing hydrogen and hydroxide ions:\(\mathrm{H_2O(l)\rightleftharpoons H^+(aq)+OH^-(aq)}\)
The key idea in this problem is the connection between equilibrium and thermodynamics. The equilibrium constant tells us how far a reaction proceeds, while the standard Gibbs free energy change tells us whether the reaction is thermodynamically favorable under standard conditions.
These quantities are related by:\(\Delta G^\circ = -RT\ln K\)
where:\(R = 8.314\ \mathrm{J,mol^{-1},K^{-1}}\)
and
\(T\) is the absolute temperature.
Because the ionization constant of water is extremely small,\(K_w = 1.0\times10^{-14}\)
we already expect the forward reaction to be unfavorable under standard conditions. Therefore, the answer should come out as a positive value of \(\Delta G^\circ\).
Concept Application
For the given equilibrium,\(\mathrm{H_2O(l)\rightleftharpoons H^+(aq)+OH^-(aq)}\)
the equilibrium constant is:\(K = K_w = 1.0\times10^{-14}\)
The temperature is:\(T = 25^\circ\mathrm{C} = 298.15\ \mathrm{K}\)
Substituting these values into the Gibbs free energy equation will directly give the standard free energy change for the autoionization process.
Solution
Using\(\Delta G^\circ = -RT\ln K_w\)
Substituting the numerical values,\(\Delta G^\circ = -(8.314)(298.15)\ln(1.0\times10^{-14})\)
Now evaluate the logarithmic term:\(\ln(1.0\times10^{-14}) = -14\ln 10\)
Since\(\ln 10 = 2.303\)
we obtain\(\ln(1.0\times10^{-14}) = -14(2.303) = -32.24\)
Substituting back,\(\Delta G^\circ = -(8.314)(298.15)(-32.24)\)\(\Delta G^\circ = 79.9\times10^4\ \mathrm{J,mol^{-1}}\)
Converting joules to kilojoules,\(\Delta G^\circ = 79.9\ \mathrm{kJmol^{-1}}\)
Therefore,\(\Delta G^\circ = +79.9\ \mathrm{kJmol^{-1}}\)
Insight
A useful relationship to remember is:
- If \(K>1\), then \(\Delta G^\circ<0\)
- If \(K<1\), then \(\Delta G^\circ>0\)
Since water has an extremely small ionization constant, \(K_w=10^{-14}\), the equilibrium lies overwhelmingly toward molecular water. The large positive value of \(\Delta G^\circ\) is simply the thermodynamic expression of that fact.
Final Answer:\(\\Delta G^\circ = +79.9\ \mathrm{kJmol^{-1}}\)