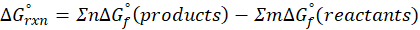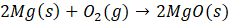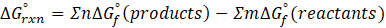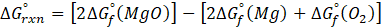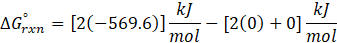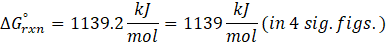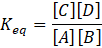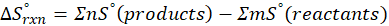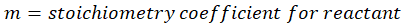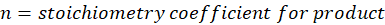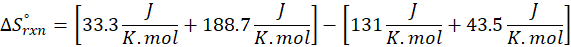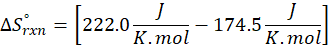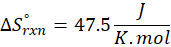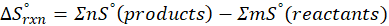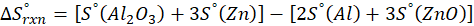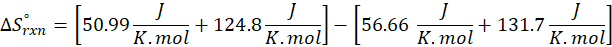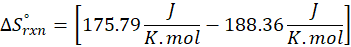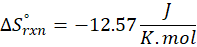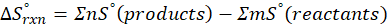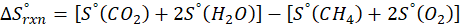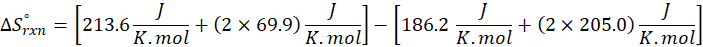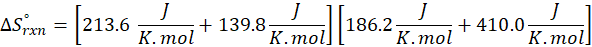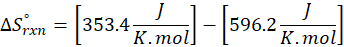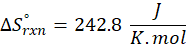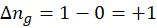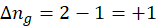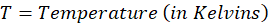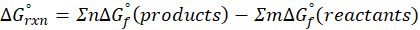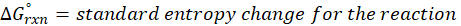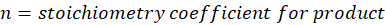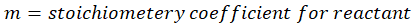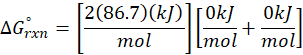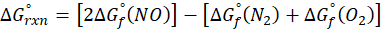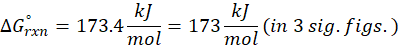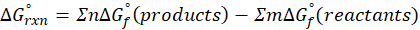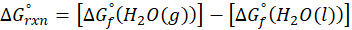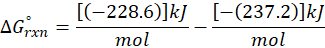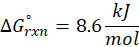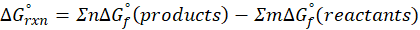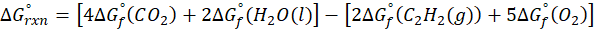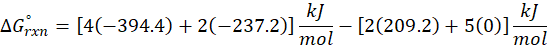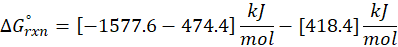Calorimetry is a technique which we use to measure the amount of heat released or absorbed during a reaction. To measure the heat of a reaction accurately, it is essential to isolate the system to avoid heat exchange with the environment. We accomplish this using a device known as a calorimeter.
Calorimeter mainly consists of metallic vessels which are good conductor of heat such as aluminium and copper etc. The vessel includes a built-in stirring mechanism to mix its contents. This vessel is placed inside an insulated jacket to minimize heat loss to the surroundings. A single opening is provided for inserting a thermometer to monitor temperature changes during the reaction.
Principle of Calorimetry
The principle of calorimetry is based on the law of conservation of energy, which states that energy cannot be created or destroyed, only transferred from one body to another. When two objects at different temperatures come into contact, the object at the higher temperature transfers heat energy to the one at the lower temperature. This heat transfer continues until both objects reach the same temperature, a state known as thermal equilibrium.
Example
Consider a scenario where someone places a hot iron rod into a container of cool water. Since the iron rod is at a higher temperature and the water is at a lower temperature, heat will transfer from the rod to the water.
As a result:
- The temperature of the iron rod decreases.
- The temperature of the water increases.
This exchange of heat continues until both the rod and the water reach the same final temperature. According to the principle of calorimetry, the total heat lost by the hot object is equal to the total heat gained by the cooler one. Mathematically, we express it as follows:
qwater + qiron rod = 0
By rearranging this gives:
qwater = – qiron rod
Here, q represent the amount of heat transferred, we calculate it using the formula:
q = mcΔT
Where:
- m is the mass of the substance (in grams),
- c is the specific heat capacity (a material-specific constant),
- ΔT is the change in temperature, calculated as:
ΔT =Tfinal – Tinitial
Where, Tfinal is final temperature and Tinitial is initial temperature.
This formula allows us to quantify the heat exchanged between substances during thermal interactions and is fundamental in calorimetry calculations.
Types of Calorimeter
- Bomb Calorimeter
- Coffee cup calorimeter
Let’s discuss about these calorimeter in detail:
- Bomb Calorimeter: A bomb calorimeter is a device used to measure the heat of combustion of a substance. It functions based on the principle of calorimetry and operates by burning a sample in a sealed chamber filled with high-pressure oxygen at constant volume. Scientists refer to this sealed chamber as the “bomb” due to its design, which can withstand the force generated during combustion. In most bomb calorimeters, water surrounds the chamber as it absorbs the heat released during combustion process.
- Coffee cup Calorimeter: This calorimeter is a simple yet effective tool for measuring heat transfer during chemical reactions in liquid solutions. It operates at constant pressure and typically consists of two nested Styrofoam coffee cups with a lid to provide thermal insulation. Laboratories frequently use this type of calorimeter due to its convenience, affordability, and suitability for basic thermochemical experiments.
Applications
- Food technologists can determine the energy content of food by measuring the heat released during combustion using calorimeter in food laboratories.
- Researchers can determine the specific heat capacity of a material by measuring the heat exchanged during temperature changes using calorimeter.
- Calorimetry is useful for analyzing medicines and other biological substances by measuring heat changes during molecular interactions, helping to understand their properties and effects.
Limitations
- Assuming the solution is pure water:
- In reactions involving aqueous solutions (like acids and bases), we often assume the solution has the same density (1 g/mL) and specific heat capacity (4.18 J/g°C) as water.
- However, if the solution contains dissolved substances (like salts or acids), its actual properties may differ, leading to inaccuracies.
2. Assuming no heat is lost to the surroundings:
- In reality, some heat is always lost to the container or environment, especially in simple calorimeters like coffee cup calorimeters.
- This causes the measured temperature change to be smaller than it would be in a perfectly insulated system, underestimating the true heat change.