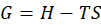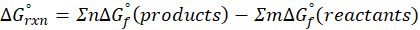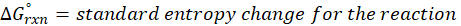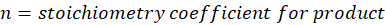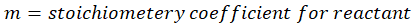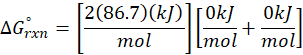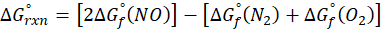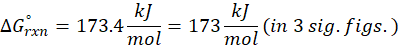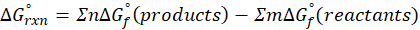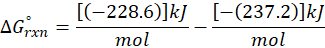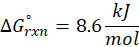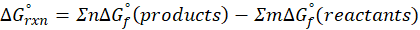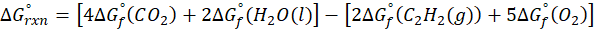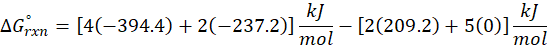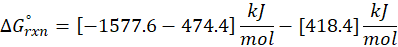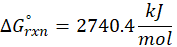The concept of Gibbs free energy was developed by the American scientist Josiah Willard Gibbs. Initially, he termed it “available energy” to evaluate the spontaneity of chemical reactions in relation to change in entropy and enthalpy within the system. We denote Gibbs free energy by symbol “G”.
Moreover, this energy is a state function, which means that it depends solely on the system’s current condition and is independent of the pathway taken to reach that condition.
Formula
Gibbs free energy is defined as the difference between the system’s enthalpy and the product of the temperature and the system’s entropy.
G = H -TS
Where,
G = Gibbs free energy
H = Enthalpy
T = Temperature
S = Entropy
Gibbs Free Energy Change
It is a thermodynamical quantity that gives the free energy at standard experimental conditions. As a result, to name the energy of a thermodynamic system as standard free energy, the reactants and products of that system should be at the standard conditions.
ΔG = ΔH – TΔS
Where, ΔG = Gibbs free energy change
ΔH = Enthalpy change
T = Temperature (in Kelvin)
ΔS = Entropy change
Gibbs free energy change and spontaneity
This energy is utilized to determine whether a reaction is spontaneous, non-spontaneous, or at equilibrium.
To proceed, we will apply ΔG formula to calculate the desired value of ΔG:
ΔG = ΔH – TΔS
Therefore, by determining ΔG, we can classify the process as spontaneous, non-spontaneous, or at equilibrium, based on the provided table.
| ΔG | Interpretation |
| ΔG>0 | Reaction is non – spontaneous (reaction is endergonic) |
| ΔG<0 | Reaction is spontaneous (reaction is exergonic) |
| ΔG=0 | The system is at equilibrium (there is no net change either in forward or reverse direction) |
Effects of delta H, delta S and T on spontaneity
| Case | ΔH | ΔS | ΔG | Spontaneity (yes/No) |
| I | + | + | +(at low T) -(at high T) | No (at low T) Yes (at high T) |
| II | + | – | + (at all T) | No |
| III | – | + | – (at all T) | Yes |
| IV | – | – | – (at low T) + (at high T) | Yes (at low T) No (at high T) |
Example:
Using the values of ΔH and ΔS, predict which of the following reactions will be spontaneous at 25°C:
Reaction A: ΔH = 10.5 kJ/mol, ΔS = 30 J/K·mol
If the reaction is non-spontaneous at 25°C, determine the temperature at which it may become spontaneous.
Solution
Interpretation: A spontaneous reaction is one that releases energy, and so the sign of ΔG must be negative. Change in free energy ΔG is defined as, ΔG = ΔH – TΔS .
We have given the reaction with specific ΔH and ΔS values at a particular temperature, we will calculate the ΔG value using the formula provided. Subsequently, this will enable us to determine the spontaneity of the reaction.
Reaction A: ΔH =10.5 kJ/mol , ΔS = 30 J/K.mol ,
Temperature = 25 +273 = 298
ΔG = ΔH – TΔS
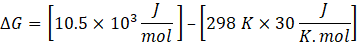
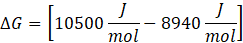
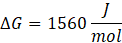
Since ΔG is positive, Reaction A is non-spontaneous. However, the reaction can become spontaneous when the temperature exceeds 25°C, as ΔG may become negative at that temperature, indicating spontaneity.
Standard state free energy change for a reaction
The standard free energy change for a reaction, represented by ΔG°rxn, is defined as the difference in the standard free energies of formation between the products and reactants. Therefore, it serves as an indicator of the reaction’s spontaneity under standard conditions.
We can calculate standard free energy change by using the following formula:
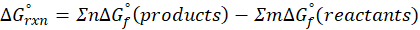
Where , ΔG°rxn = Standard entropy change for reaction
n = stoichiometry coefficient for product
m =stoichiometry coefficient for reactant
∑ = sum of
Example: For reaction :
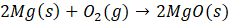
In order to find ΔG°rxn , we will use this formula:
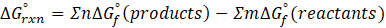
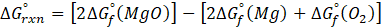
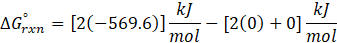
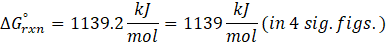
Therefore, ΔG°rxn for the reaction is 2 Mg (s) + O2 (g) → 2 MgO(s) is 1139 kJ/mol.
Relation between Gibbs free energy (delta G) and Equilibrium constant (K)
Let’s consider the following reversible reaction as
A + B ⇌ C + D
The relation between delta G and equilibrium constant for the given reaction as:
ΔG=ΔG°+RTlnQ
Where, ΔG = Free energy
ΔG° = Change in standard free energy
R = ideal gas constant ( 8.314 J/mol.K)
T = Absolute temperature in Kelvin
Q = reaction Quotient
At Equilibrium ΔG = 0, Q = Keq
[ Keq for above reaction is :
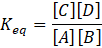
So, the relation at equilibrium is:
0 = ΔG° + RT ln Keq
ΔG° = – RT ln Keq
Consequently, The equation ΔG° = – RT ln Keq , describes the relation between ΔG°(standard free energy change) and Keq (Equilibrium constant). Specifically, R = Gas Constant and T = Absolute Temperature.
In chemistry, this equation is a fundamental relation in thermodynamics, as it allows us to determine the equilibrium constant of a reaction when the change in standard free energy is known, and vice versa.